Carbon dioxide in water, can be free of carbon dioxide gas (CO2), the bicarbonate ion (HCO 3), carbonate ion (CO32-) and carbonic acid (H2CO3). This free carbon dioxide needed in photosynthesis by plants berhijau daun.Sedangkan carbonate and bicarbonate salts, especially salts of calcium needed to support the water pH. Fused carbon dioxide with water to form carbonic acid (H2CO3) that produce acidic conditions in water into H + and HCO3- reactions are as follows:
![]()
a) Sources of carbon dioxide.
Carbon dioxide contained in the water can be obtained from:
diffusion from the atmosphere directly
groundwater through the soil organic
Rainwater, rain falling to the earth’s surface theoretically has a carbon dioxide content of 0.55 to 0.6 mg / l
The results of the decomposition of organic material in the bottom waters
From the results of the process of breathing (respiration) animals and water plants,
The results of the process of solving / decomposition of chemical compounds.
b) The decline in carbon dioxide in the water.
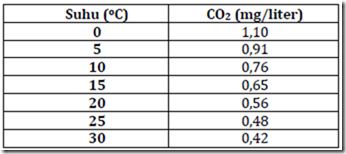
As with other faktorkimia, carbon dioxide solubility is influenced by temperature, pH and carbon compounds. The solubility of carbon dioxide in the water can be seen in Table 5.
Table 6. Effect of temperature on the solubility of carbon dioxide of natural waters
Influence of carbon dioxide on the lives of aquatic organisms can be direct (respiration) or indirect (photosynthesis). In general, the effect of carbon dioxide on aquatic organisms are as follows:
a) In the range of 15 ppm will affect the lives of fish (aquatic organisms) because it is toxic to the organism.
b) Required by berhijau plant leaves (chlorophyll) for photosynthesis.
c) to maintain a stable pH in the water, especially in the form of carbonate / bicarbonate. This means the water can maintain a stable environment to support the life of the organism.