To determine the amount of the total concentration of ammonia nitrogen in water sample used spektrofotomerik principle conducted in labs. In order to be readable by a machine spectrophotometer, ammonia in 10 ml water sample that has been filtered to be treated beforehand with 0.5 ml phenol and 0.5 ml of sodium nitroprusside were homogenized, and then the reaksikan back with oxidizing reagent 1 ml and homogenized back.
Thereafter, the reaction tube used to conduct the reaction was sealed and allowed to stand for one hour. Then the color of the water sample absorbance (blue) are measured with a spectrophotometer at a wavelength of 640 nm. The blue color is caused due to formation of compounds indofenol. Then the absorbance of water sample absorbance adjusted with distilled water (blank) and a constant calculation (Stirling et al., 1985dalam Subandiyo, 1992).
Ways of working :
(1) Filter 25-50 ml water sample with Whatman filter paper no 42 (do not use a vacuum pump, so that no ammonia is lost.
(2) Pipette 10 ml water sample was filtered, put in a glass cup
(3) While stirring with a magnetic stirrer add 1 drop mnso4, chlorox 0.5 ml, and 0.6 ml phenate. Let stand for ± 15 minutes, until the formation of stable color (the color will remain stable for several hours)
(4) Create a blank solution of 10 ml of distilled water, perform the procedure 3
(5) Create a standard solution of 10 ml of standard solution of ammonia (0.3 ppm) to do the procedure 3
(6) With reference solution at a wavelength of 630 nm, spectrophotometer set at 0.00 absorbance (transmitteance 100%) and then take the measurement sample and the standard solution
(7) Record the data obtained
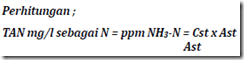
Information:
Cst: the concentration of the standard solution (0.03 mg / l)
Ast: the value of absorbance (transmittance) standard solution
As: absorbance value (transmittance) of water samples
The measured concentration of the factors stated in the levels of nitrogen (N) contained in ammonia (NH3). To determine the concentration of ammonia NH3 are expressed in mg / L, the value above TAN multiplied by actors such as the following equation:
![]()
Information :
BM: molecular weight
BA: atomic weight