Ion exchange is a process in which ions adsorbed on a surface of the filter media exchanged with other ions that are in the water. This process is made possible through a phenomenon of attraction between the surface of the media charged with polar molecules. When a charged molecule touching a surface that has the opposite charge of the molecule will be chemically attached to the surface of it.
First cations with a valence greater will be exchanged prior cations with smaller valence. For example, if there is iron in the water (air-valence 3), calcium (ber- valence 2) and ammonium (ber- valensi1) in the same amount, it will first iron sequestered by the zeolite, following last calcium and ammonium.
Second, the cation concentration is highest in the water will be sequestered in advance although smaller valence. For example, in the above case, when the concentration (amount) of ammonium far more than the iron and calcium, then in accordance with rule 2, ammonium jerap be inclined at first.
By the processes mentioned above, the chemical filter can be used to “purify” the water of paritkel molecular-sized particles that can not be processed mechanically or biology. Some things that can be removed with a chemical filter include toxic effects, hardness, color and dissolved organic particles.
Figure 2. Activated charcoal, Resin and Zeolite
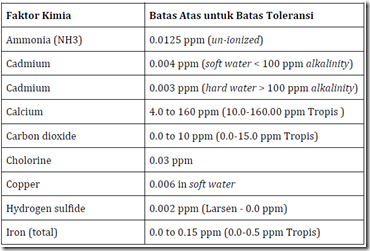
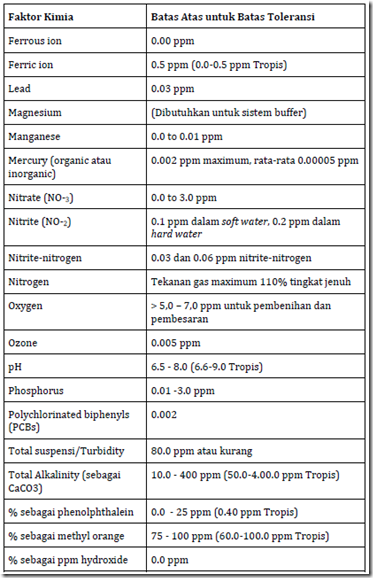
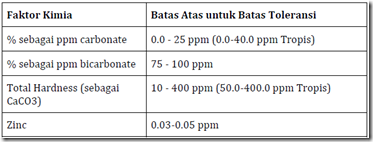
The range of chemical parameters of water quality varies in different designation, such as for drinking water will be different to that used for aquaculture. The table below is a range of chemical parameters of water quality for aquaculture
Table 4. Chemical parameters of water quality and the value of upper and lower limits are recommended for seeding or enlargement of fish / shrimp. The concentration is in ppm (mg / 1). (Source: Modification from Wedemeyer, 1977; Piper, etc al. (Larsen), 1982)